Progressive muscle weakness remains the clinical hallmark of ALS, but there is no ideal muscle biomarker.
There is a major unmet need for quantitative tools that quantify muscle degradation and assess muscle quality in clinical settings with minimal patient and provider burden.
Why is ALS Diagnosis So Hard?
ALS begins insidiously in a single muscle. Progression is fast, varies by person.
![]() Two people with ALS can have extremely different presentation, in totally different body regions.
Two people with ALS can have extremely different presentation, in totally different body regions.
![]() ALS begins within single muscles, somewhere in one body region.
ALS begins within single muscles, somewhere in one body region.
![]() Variation in upper vs. lower motor neuron involvement contributes to additional diagnosis inconsistency
Variation in upper vs. lower motor neuron involvement contributes to additional diagnosis inconsistency

![]() Little agreement as to what muscle measures to use, in each clinical trial, often occurs.
Little agreement as to what muscle measures to use, in each clinical trial, often occurs.
![]() Remarkable variability in the speed of ALS progression.
Remarkable variability in the speed of ALS progression.
![]() Highly variable ALS progression can lead to unrealistic, insufficient patient sizes for clinical trials.
Highly variable ALS progression can lead to unrealistic, insufficient patient sizes for clinical trials.
ALS Clinical Trial High Failure Rate
There are four key reasons why:
1. Clinical trials have tended to include ALS patients as though this is a homogeneous, rather than a heterogeneous disease.
2. The lack of a consistent, quantitative, comprehensive measure of disease progression is a significant issue for clinicians.
3. The high variability in the speed of disease progression, making it possible that target effect sizes are unrealistic.
4. Slow time to diagnosis in ALS can mean that the disease process has progressed to a stage where preserving motor neuron health may be particularly challenging.
The ability to quantify muscle condition accurately and with high reproducibility is critical if we are to rapidly diagnosis individuals with neuromuscular conditions, plan treatment and therapy, and track the progression and effects of therapy over time.
Especially for ALS. It moves too fast.
Fighting ALS.
What if there was a way to…

Breakthrough ALS Clinical Research
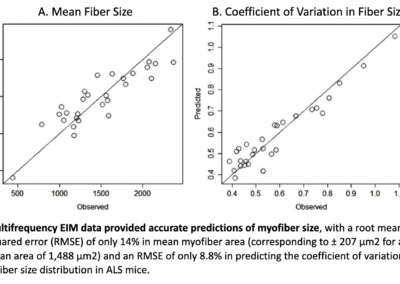
The main feature of muscle diagnostic relevance is myofiber atrophy
EIM has the unique ability to provide “virtual biopsy-type” data, showing high concordance between observed vs. predicted values.
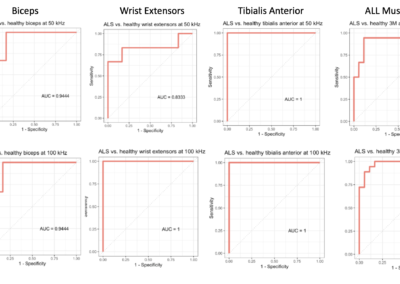
The Myolex EIM impedance-based approach is very sensitive to deteriorating effects of injury and disease.
EIM has demonstrated that it can provide predictive variables to assess myofiber size and distribution with good accuracy, particularly in diseases where myofiber atrophy is the predominant histological feature.
Importantly, this can be done non-invasively, without the requirement for biopsy or burdensome quantification. Because EIM provides spectral data on muscle composition, deep learning techniques, extracting all information buried within the impedance data set, can then be leveraged to optimize EIM’s ability to assess muscle fiber size, contractility, and composition (Ref. 8).
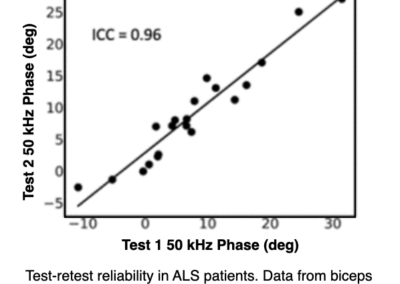
EIM is highly reliable and reproducible
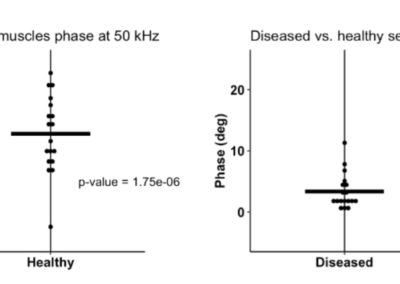
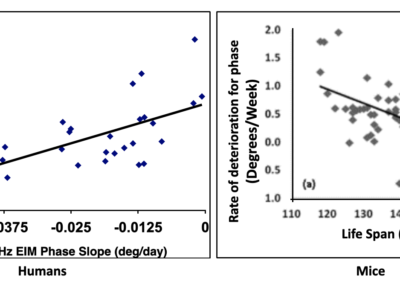
EIM. Significant correlation between 50kHz phase value and ALS patient survival
In an ALSA-funded studies, EIM showed substantial sensitivity to ALS disease status in mice and humans, and significant correlation between decline in 50 kHz phase value and ALS patient survival (shown here in years).
This data not only demonstrates EIM’s predictive capability, but it also provides construct validity that EIM is evaluating something deeply meaningful.
The slower the rate of EIM 50 kHz phase decline, the longer the survival of the patient (Ref. 14).
Smaller, faster clinical trials? It's now possible
EIM offers substantial flexibility in terms of its implementation. One can tailor study to a single body region which is most affected by ALS, an approach that cannot be used readily with other disease-tracking tools.
This could translate into tracking a single limb that is most rapidly progressing from the disease during a clinical trial rather than adopting a far less sensitive assessment of whole-body decline.
By performing such directed measurements, it becomes possible to detect a treatment effect over shorter periods of time and with fewer subjects, helping to facilitate and speed the completion of clinical trials.
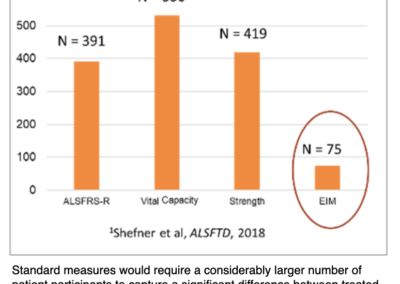
EIM has the potential to serve as a powerful, future biomarker in ALS clinical trials that will be able to reduce clinical trial sample size requirements dramatically (Ref. 3).
Frequency reduces sample sizes
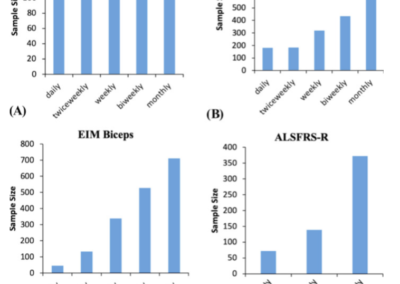
The effect of more frequent muscle measurements results in smaller sample size estimates.
For most measures, there is an increase in effect size when more frequent measurements are made. This results in a marked reduction in sample size, in part, because sample size is inversely related to the square of the effect size.
Frequent at-home measurements, using tools such as EIM, holds the prospect of tracking progression more frequently, thereby reducing sample size requirements for clinical trials in ALS while also being acceptable to the patients (Ref. 12).
ALS patients can feel more in control at-home
The Patient Perspective.
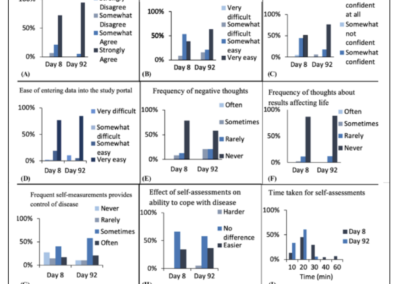
One of the key objectives of this The ALS At Home study was to determine whether participants with ALS or their caregivers could be trained to assess their clinical status by performing a variety of outcome measures at home (Ref. 12).
Conclusions:
- Participants generally felt that they had improved in their ability to take measurements over time, with most completing the entire set of measurements within 20 min by 92 days.
- Of the ALS participants who persisted in measurement, most indicated that the study actually made them feel more in control of their disease.
- The generally positive reactions to home assessments could indicate greater engagement and persistence, as well as a greater sense of personal responsibility.
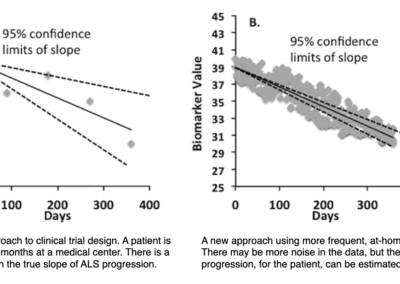
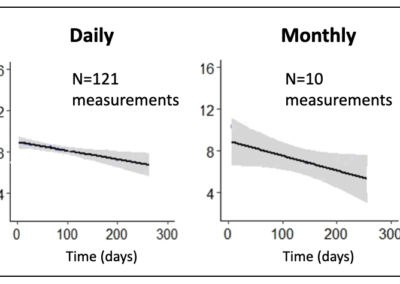
More frequent measurements at-home, greater certainty in ALS progression
Daily, at-home measurements leads to more accurate tracking
This figure shows data that demonstrates greatly improved accuracy in quantifying the rate of decline in one patient’s biceps. This improved accuracy means better tracking of individual patients and clinical trials with fewer subjects that can be done at home.
Daily, at-home measurements could be performed with a greater 10X improvement over monthly measurements in terms of reductions in sample size (Ref. 5).
Let’s Discover Together.